Bacteriophages, the friendly viruses which will save us from ever-increasing antibiotic resistance
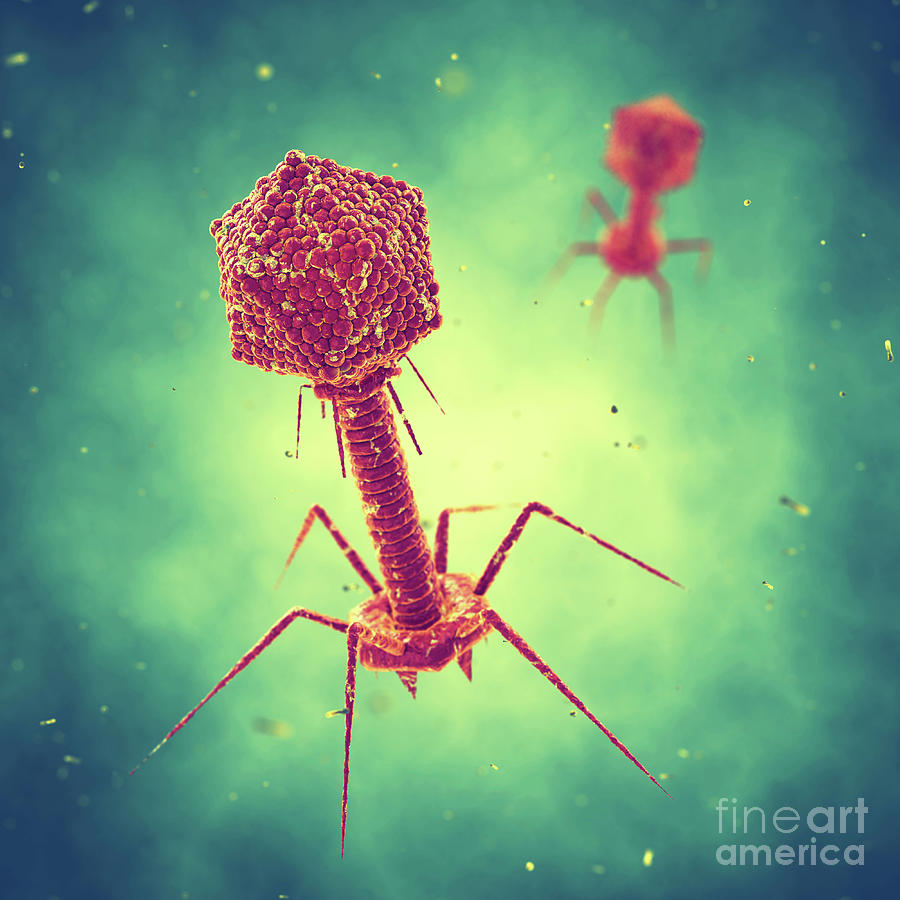
Bacteriophages, the friendly viruses
Just a week ago, a new order from Jarrow Formulas arrived which contained an excing new product, called 'Immune Booster'' . Actually, I wasn't even aware how exciting it is, since I simply decided to order Jarrow's newest product without fully checking all of its details.
Nonetheless, when we did check the information on Jarrow's own website, it was still shrouded in mystery: Immune Booster was called a prebiotic, while it contains both a beneficial probiotic 'Bacillus subtilus' bacteria as well as a registered trademark ingredient called 'PreforPro®' which contains 4 different 'ingredients'c alled 'viridae'. "Viridae?" , isn't that a derivate of a virus ? And indeed it is. What's more: these 4 'viridae' are a very specific type of viruses! They are 'bacteria-killers with the generic name 'bacteriophages' where 'phage' refers to the act of 'eating'. So it's a partial probiotic due to the Bacillus, but one can't call viruses prebiotics nor postbiotics, can we?
Anyhow, the discovery Immune Booster contains bacteriophages nearly made me jump out of excitement! Why? I'd read about bacteriophage treatment in Eastern European countries, more specifically in Russia. The function of bacteriophages is to treat bacterial infections, when antibiotics are not available.
In Western countries, this habit to treat bacterial infections was also commonly used, ... until the discovery of antibiotics when it died out fairly rapidly, until, massive antibiotic resistance reared its ugly head.
This topic is also quite dear to my heart since i had a massive case of food poisoning almost 20 years ago which landed me in hospital for well over 3 weeks. Fortunately the infection could be treated with heavy-duty antibiotics.
So , without further ado, you are about to learn all you may ever want to know about bacteriophages.
Fighting fire with fire: killing bacteria with virus
Our time with antibiotics is running out. Ever more often, people die from untreatable bacterial infections because of antibiotic resistence. Bacteria have been evolving to resist antibiotics faster than a new antibiotic can be developed and approved, a process which generally takes 10 years or more.
Our slow response means that we are losing in this antibiotic arms race. We urgently need an alternative method to fight bacterial infection. One promising method for killing bacteria is to use bacteriophages: viruses that infect and kill bacteria.
Bacteriophages: Natural enemies of bacteria
Bacteriophages, called phages for short, were discovered independently by Frederick Twort in 1915 and Félix d’Herelle in 1917, over a decade before penicillin, the most well known antibiotic. In the following years, phages were employed to treat dysentery and cholera with success. These phages were isolated from the stool of patients who unexpectedly recovered from the illness. Scientists speculated that there was something in these lucky patients that helped to remove harmful bacteria from their guts. They isolated phages from the stools, purified them, and gave the phages to other patients. In one study, 63% of untreated people suffering from cholera in Punjab, India died while only 8% of those who were treated by phages died. Despite the early success, phages therapy was eclipsed by the discovery of penicillin and the rise of antibiotics.
At the time phages were initially used for treatment of cholera, scientists had only just begun to study viruses and speculate about how phages work. It was not until 1940 that the first images of phages were obtained using an electron microscope. We now know that phages are viruses that infect only bacteria. As a type of virus, phages cannot live and reproduce alone. Viruses need to invade a host cell, consume the host’s nutrients to make more copies of themselves, and lastly get out of the host cell – often by killing the host in the process.
In general, phages start their killing first by recognizing and landing on a bacteria. Each type of phages has a specific landing pad. The phage then injects its DNA into the bacteria. This DNA copies itself, makes more of the phage’s shell, and packages the newly made DNA into the new shell. Lastly, the phage produces toxic chemicals that rupture the bacterial host from inside out, releasing its newly made children to the outside to infect even more bacteria .
How phage kills bacteria.
(1) Phage first lands on bacteria.
(2) It then injects its DNA inside bacteria.
(3) The DNA is copied and is used to make packaging for a new generation of phages.
(4) Lastly, the new phages assemble and burst of bacteria, killing it in the process.

Advantages of phages over antibiotics
An antibiotic is a chemical that kills bacteria. It does so by disrupting one or more of the important processes that bacteria need to survive. Because these processes are common in many bacteria, one “broad spectrum” antibiotic could potentially kill many species of bacteria at once. While antibiotics have revolutionized medicine and are often very effective in stopping bacterial infection, well-developed phages could have several advantages over antibiotics.
First, phages are specific to one species of bacteria and are therefore unlikely to disturb beneficial microbe living in our guts. The human body is populated by over a thousand species of microbes, which are estimated to make up about 3-5 pounds of our total body weight. These microbes do important jobs for us, such as helping us make nutrients we cannot make ourselves. Because many antibiotics kill bacteria indiscriminately, treating an infection with an antibiotic results also in killing this beneficial gut bacteria. Each phage, on the other hand, evolved to kill just a specific set of bacteria. Because phage kills with a narrow scope, it could be used to cure an infection without disturbing the community of beneficial bacteria in our body.
Second, phages are able to kill antibiotic-resistant bacteria. The way that phages kill bacteria is harder for bacteria to develop resistance against compared to the way that antibiotics kill bacteria. Rather than stopping bacteria from doing one specific process like in the case of antibiotics, phages actively destroy the bacteria’s cell wall and cell membrane and kill bacteria by making many holes from the inside out. In addition, many bacteria develop biofilm – a thick layer of viscous materials that protect them from antibiotics. Many phages are equipped with tools that can digest this biofilm.
Why aren’t phages used?
With the exception of treatment options available in a few countries, phages have been largely abandoned as a treatment for bacterial infection. One main reason is because antibiotics have been working well enough over the past 50 years that most countries have not re-initiated a study on the clinical uses of phages. But another reason is that there are some limitations for using phages as a treatment.
First, phages are more difficult to prepare cleanly. To produce phages, first scientists have to grow a large quantity of bacteria that is the natural host of the phage. The bacteria is then infected with the phages, and the phages in turn reproduce and kill all the bacteria. The difficulty begins with the isolation of live phages from a multitude of dead bacteria corpses. If not removed from the final medication given to the patient, dead bacteria bodies could trigger a deadly immune response called sepsis. Another challenge is to obtain the right concentration of phages since its concentration can’t be measured directly. If the concentration is too low, phage therapy would inefficacious. Many of the early commercial phage products were of poor quality and incapable of treating infectious disease, leading to phage therapy being discredited.
Second, phage takes a longer time to employ in a treatment compared to antibiotics. Because a single type of phage can only infect a few species of bacteria, phage selection has to be done with care. First, doctors have to figure out the identity of bacteria that is causing the illness. Then they have to check whether the available phages could kill this strain of bacteria. If not, they have to search for new phages that could do the job. This process takes time that the patients may not have – especially when phages are used only as a last resort on very ill patients. On the other hand, because antibiotics kill indiscriminately, doctors can prescribe an antibiotic to treat a patient without needing to first identify the specific type of bacteria.
Other concerns about phage therapy are centered on its safety and efficacy. Because the western world abandoned phage therapy many decades ago, there is little data about these topics available. However, research on phage therapy continues and prospers in France and eastern European countries, especially in Georgia. From their studies, phage therapy does not exhibit any major safety concerns.
This specific article allowed for comments, among which one (made in August 2018) stood out
"A very successful laboratory in the eastern block [Tbilisi , Georgia] had isolated phages that were able to treat almost all bacterial infections. When the eastern block dismantled, the laboratory opened all its secrets in the hope that the research could continue. with foreign investment.
However no one was interested primaraly because the Drugs companies could not make any money out of it .
It is scandalous that many have died, who could have been succesfully treated with phages, because because of the Wests money orientated drugs culture.
For those who have untreatable bacterial infections , many can be treated by the instute in Georgia where research continues."
A BBC-documentary well worth watching : Antibiotic Resistance - The Virus that Cures - BBC Horizon
But let's continue to hear more about the wonders of bacteriophages.. one other discovery is how not just bacteria are abundant inside our body, but so are funghi and viruses! Even yours truly with university courses on microbiology and virology under her belt, had no idea!
So here's yet another more recent article written in 2020 focusing on the abundance of viruses in our gut!
How 'good' viruses may influence health
Although the role of “good” viruses in human health is still relatively mysterious, we are slowly unraveling the importance of our viral visitors. In this special feature, we introduce a neglected section of the microbiome — the virome.
The role of bacteria and our microbiome in health and disease is at the forefront of medical research.
We are a long way from answering the many questions posed by recent findings, but it is now firmly established that without our personal fleet of “friendly” microorganisms — our microbiome — we would not thrive.
Medical science, however, does not sit on its haunches; its eyes are always fixed on the horizon, straining to describe the shape of things hidden in the far distance.
As we struggle to unpick the almost unbearably complex interactions between bacteria and health, the next challenge is already waiting in the wings: the role of the virome.
What is the virome?
When we hear the word “microbiome,” we immediately think of bacteria, but technically, the microbiome is the sum of all microorganisms in a particular environment. Some scientists use the term to refer to the sum of the genetic material of these microorganisms.
So, aside from bacteria, the microbiome also includes viruses (the virome) and fungi (the mycobiome), among other visitors. To date, scientists have paid comparatively little attention to the virome or mycobiome.
Viruses have made themselves at home in a range of ecological niches in the human body, especially on mucosal surfaces, such as the insides of the nose and mouth and the lining of the gut.
In this feature, we will concentrate on the gut virome because it hosts the greatest number of viral occupants and has been investigated the most.
Of course, viruses are most famous for causing diseases, such as smallpox, hepatitis, HIV, and rabies. Because of the urgency associated with viral disease, this aspect has taken up the lion’s share of researchers’ time. However, many viruses do not have the slightest interest in human cells.
Introducing the bacteriophage
Scientists consider the virome to be “the largest, the most diverse, and the most dynamic part of [the] microbiome,” and the majority of the viruses in our guts are bacteriophages. Wherever there are bacteria, there are bacteriophages in abundance.
As other researchers explain: “Phages are the most abundant life forms on Earth, being virtually omnipresent. […] Some freshwater sources may contain up to 10 billion per [milliliter].”
Bacteriophages infect bacteria, commandeer their cell machinery, and use it to replicate their genetic material.
It is now abundantly clear that gut bacteria influence health and disease, so it is no surprise that viruses that infect gut bacteria may have a significant influence, too.
Phage therapy
From the 1920s to the 1950s, scientists investigated whether bacteriophages could be used to treat bacterial infections. After all, these viruses are adept at destroying human pathogens.
Scientists found that phage therapy was both effective and, importantly, free from side effects.
When antibiotics were discovered, phage therapy faded into the background. Antibiotics could be manufactured with relative ease, and they killed a broad spectrum of bacterial species.
However, with today’s hi-tech capabilities and the fearsome backdrop of antibiotic resistance, interest in phage therapy may enjoy a resurgence.
One factor that makes phage therapy attractive is its specificity. Often, antibiotics wipe out a wide spectrum of bacterial species. Now that we know that “good” bacteria live in the gut, however, it is clear that this is not ideal.
Bacteriophages, meanwhile, only target a narrow range of strains within the same bacterial species.
Plus, they only replicate if their target bacteria are in the local area. Taken together, this means that they only attack the desired bacterium, and they continue to replicate until they have wiped out the infection.
Friends for life
Bacteriophages join the human journey at an early stage. One study examined meconium — a newborn’s first poop — and found no evidence of viruses.
However, just 1 week after birth, each gram of a baby’s poop contained about 100 million virus particles, most of which were bacteriophages. Our virome truly is a lifelong companion.
Each human has a distinct selection of bacteriophages, which is collectively referred to as the phageome. People who have roughly the same diet share more similarities, but overall, each individual’s phageome varies wildly.
From symbiosis to dysbiosis
Bacteriophages, as mentioned, destroy bacteria. However, in some situations, bacteriophages can benefit populations of bacteria.
In the gut, bacteriophages predominantly exist as prophages. In this stage, their genetic code is incorporated into a bacterium’s genome, ready to produce bacteriophages if activated.
At this point in their life, a bacteriophage is not harmful to a bacterium — they exist in symbiosis.
Because bacteria can exchange genetic material with each other, the genetic code of prophages can also be transferred between individual bacteria.
They can exchange “genes associated with antibiotic resistance, virulence, or metabolic pathways between different bacterial species.” This could benefit some bacterial species, potentially allowing them to broaden their niche. However, the growth could be at the expense of other colonies of bacteria in the gut.
“Prophages are symbiotic to their host bacteria, and these bacteria are symbiotic to our body. Therefore, phages can indirectly provide benefit to a multicellular organism like [a] human beyond what is experienced immediately by their host bacterial cells.”
Once prophages are triggered to become active — for instance, in times of stress or if the host bacterium is in danger — they can cause a widespread change in the gut’s microbial community.
The shift from harmless prophage to so-called lytic phage can wipe out communities of bacteria, potentially providing “bad” bacteria with some breathing space and allowing them to fill the void.
This is called community shuffling and can lead to dysbiosis — a microbial imbalance.
From dysbiosis to diagnosis
Dysbiosis is associated with a range of conditions, including inflammatory bowel disease, chronic fatigue syndrome, obesity, Clostridium difficile infection, and colitis. However, researchers are still unsure of the role of bacteriophages in these conditions.
In these cases, dysbiosis might occur via other mechanisms. Alternately, it might be a symptom of the conditions, rather than the cause.
Researchers have observed changes in gut bacteria in a surprisingly varied range of diseases, including type 2 diabetes, schizophrenia, depression, anxiety, Parkinson’s disease, and many more.
Because bacteriophages outnumber the bacteria in our guts and rely on them to replicate, they must be either affected by or involved in any fluctuations.
Bacteriophages may not be driving changes in the gut — changes that, it must be added, may not be driving the disease. Instead, bacteriophage populations might just be altered, passively, by the changes in gut bacteria.
Whether the ebb and flow of bacteriophage communities is important in health and disease will be challenging to investigate. But even if it is not pivotal in the pathology of a disease, spotting these fluctuations might have other benefits.
As an example, there is the potential to use the virome as a diagnostic marker. For instance, scientists have identified disease-specific alterations in the gut virome in people with inflammatory bowel disease, which is a notoriously difficult condition to diagnose.
The trouble with viruses
Studying bacteria is far from easy; after all, they are incredibly small. Bacteria are generally 0.4–10 micrometers across. To provide some context: 10 micrometers is just one-hundredth of a millimeter or four ten-thousandths of an inch.
Viruses, however, are even smaller, at just 0.02–0.4 micrometers across.
Aside from the difficulties inherent in working on such a tiny scale, viruses pose other challenges.
If scientists want to understand which bacterial species are present in any given population, they extract genetic information.
From this, they isolate specific stretches of code and match them to existing databases; most commonly, they use the 16S rRNA gene. This particular gene can be found in almost all bacterial species, and over evolutionary time, it has remained relatively unchanged.
However, some regions of 16S RNA are considered hypervariable. Differences between these regions allow researchers to identify species.
Viruses, on the other hand, do not share any equivalent genes among species. This, until relatively recently, made studying the virome almost impossible, but advances in next-generation sequencing are slowly knocking down barriers.
At this stage, the role of viruses in human health is nowhere near as clear as their role in disease.
With that said, it also seems highly likely that viruses do play a substantial part in maintaining a healthy body. Only with advances in research techniques will their full impact be understood.
Given the immediate concerns of antibiotic resistance, perhaps renewed interest in the bacteriophage will see more time dedicated to this mysterious element of medical science.
Still, understanding the interplay between the components of our microbiome will be hard-won information; as one paper explains:
“The composition of the gut microbiome is not the same during the several stages of life, or even during the hours of the same day.”
This is sure to be a long battle.
Press release on Immune Booster
Jarrow Formulas®, the #1 probiotic brand for customer satisfaction, announced the launch of Immune Booster, formulated with two clinically studied and supported ingredients. The new product supports a healthy immune system and normal digestion.
Available in an easy, single serve stick pack, Immune Booster offers a tasty, orange-flavored, fast-melt powder that quickly dissolves on the tongue without the need for water.
"Science has clearly demonstrated that 70% of the immune system is housed in the gut, so we knew in developing Immune Booster, we wanted to focus on the microbiome as the immunity power center," said Hanan Wajih, Chief Marketing Officer of Jarrow Formulas. "Our goal was to create something that could conveniently fit into people's morning lifestyles and routines while adhering to Jarrow's strict wellness and efficacy standards."
Made with PreforPro®, a rockstar prebiotic that helps balance the gut microbiota by allowing beneficial probiotic bacteria to have more room to flourish, Immune Booster provides bacteriophages. Abundant in nature, phages are submicroscopic bundles of DNA and RNA with a protein shell that are found in seaweed, soil, fermented foods and in the human body. Able to function in both the small and large intestines, bacteriophages help support gut health.
In addition to PreforPro®, DE111® is another clinically supported ingredient in Immune Booster that functions as a spore-forming, immune-supporting probiotic strain.
DE111® works by secreting digestive enzymes, biosurfactant and exopolysaccharides to support a healthy gut and immune system.
Always focused on the customer and their health concerns, Immune Booster comes at a time when immunity is a top priority for consumers. A nationwide survey by Jarrow found that immunity is the #1 specific benefit consumers look for, and 81% of consumers have used or are interested in taking a vitamin or supplement with immune benefits.
Jarrow Formulas® strives to meet the needs of all consumers with all products. Immune booster is Non-GMO, vegetarian and contains no wheat, gluten, soybeans, dairy, egg, fish/shellfish, peanuts/tree nuts or sesame.
Recent posts
- Better fit and fat than lazy and thin? Fit at every size, yet 'fat but healthy’ is a myth!
- Gut health and sleep: can your microbiome keep you up at night?
- Is chewing gum bad for your health? Chewing gum exposes us to microplastics and may ruin dental health
- Radiation from wearable devices, is it a reason for concern?
- Diabetes and heart disease , how they are related and what can you do about it?
- Acute versus chronic pain: what's the difference and how to get rid of it or learn to cope with it?
- How common are spinal or foot abnormalities?
- Why are fertility rates declining around the world?
- What virus have I been affected by? What is the best way to deal with a viral infection?
- On anosmia, the loss of your sense of smell and how to regain the sense of smell naturally


